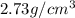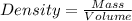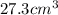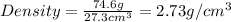Chemistry, 31.08.2019 10:00 allicat133
Calculate density of aluminum if 27.6cm^3 has a mass of 74.6g

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 16:00
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3

Chemistry, 23.06.2019 01:00
How does carbon monoxide pose the greatest threat to humans? a. it can be produced by wood fires. b. it can be produced by home furnaces. c. it is produced by acid rain. d. it is produced by modern automobiles.
Answers: 2
You know the right answer?
Calculate density of aluminum if 27.6cm^3 has a mass of 74.6g...
Questions

Mathematics, 03.02.2021 16:30

Mathematics, 03.02.2021 16:30

English, 03.02.2021 16:30

Mathematics, 03.02.2021 16:30



English, 03.02.2021 16:30

English, 03.02.2021 16:30

Mathematics, 03.02.2021 16:30

Mathematics, 03.02.2021 16:30

Mathematics, 03.02.2021 16:30

Mathematics, 03.02.2021 16:30

Chemistry, 03.02.2021 16:30


Mathematics, 03.02.2021 16:30


History, 03.02.2021 16:30

Advanced Placement (AP), 03.02.2021 16:30

Mathematics, 03.02.2021 16:30

Social Studies, 03.02.2021 16:30