
Chemistry, 15.10.2019 12:00 runaway173
For the n = 3 electron shell, which of the following quantum numbers are valid? check all that apply.
l = 3
m = 3
l = 0
m = –2
l = –1
m = 2

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1


Chemistry, 22.06.2019 11:20
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1

Chemistry, 23.06.2019 01:00
Which of the following is the molecular formula for a simple sugar? a. cooh b. h2o c. oh d. c6h12o6
Answers: 1
You know the right answer?
For the n = 3 electron shell, which of the following quantum numbers are valid? check all that appl...
Questions


Mathematics, 01.07.2019 16:30

English, 01.07.2019 16:30

English, 01.07.2019 16:30


Biology, 01.07.2019 16:30

History, 01.07.2019 16:30

History, 01.07.2019 16:30

Biology, 01.07.2019 16:30

History, 01.07.2019 16:30


Health, 01.07.2019 16:30




History, 01.07.2019 16:30

Biology, 01.07.2019 16:30


English, 01.07.2019 16:30

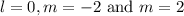
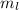 . The value of this quantum number ranges from
. The value of this quantum number ranges from 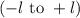 . When l = 2, the value of
. When l = 2, the value of 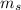 The value of this is
The value of this is 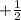 for upward spin and
for upward spin and 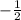 for downward spin.
for downward spin.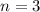
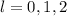
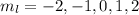
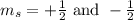 (For each sub-shell)
(For each sub-shell)

