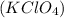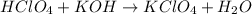Chemistry, 21.09.2019 09:30 Victoriag2626
What acid and what base would you choose to prepare the salt potassium perchlorate (kclo4)?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2


Chemistry, 23.06.2019 07:20
F1.5 mol of nabh4 react, how many moles of b2h6 are formed? 2 nabh4(aq) + h2so4(aq) → 2 h2(g) + na2so4(aq) + b2h6(g)
Answers: 1
You know the right answer?
What acid and what base would you choose to prepare the salt potassium perchlorate (kclo4)?...
Questions

History, 04.02.2020 00:08

Physics, 04.02.2020 00:08

Health, 04.02.2020 00:08


Biology, 04.02.2020 00:08

Biology, 04.02.2020 00:08

History, 04.02.2020 00:08

History, 04.02.2020 00:08

History, 04.02.2020 00:08

English, 04.02.2020 00:08





Mathematics, 04.02.2020 00:09

Mathematics, 04.02.2020 00:09


Mathematics, 04.02.2020 00:09