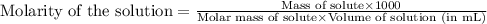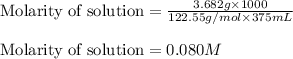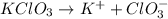Chemistry, 01.09.2019 07:30 jhenifelix
A3.682 g sample of kclo3 is dissolved in enough water to give 375. ml of solution. what is the chlorate ion concentration in this solution?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 21.06.2019 18:30
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible?
Answers: 2

Chemistry, 22.06.2019 03:30
Each pair of clay balls represents to planetesimals if each plane test molluscum pound of the same material and is separated by the same distance which pair experiences the greatest gravitational attraction
Answers: 2

Chemistry, 22.06.2019 09:40
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1
You know the right answer?
A3.682 g sample of kclo3 is dissolved in enough water to give 375. ml of solution. what is the chlor...
Questions

History, 24.02.2021 01:00

English, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00


History, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00




Spanish, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00

History, 24.02.2021 01:00

German, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00

Chemistry, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00