
Chemistry, 18.09.2019 02:00 hchxxhBfncndnd9319
The haber process can be used to produce ammonia (nh3) from hydrogen gas (h2) and nitrogen gas (n2). the balanced equation for this process is shown below.
3h2 + n2 mc025-1.jpg 2nh3
the molar mass of nh3 is 17.03 g/mol. the molar mass of h2 is 2.0158 g/mol. in a particular reaction, 0.575 g of nh3 forms. what is the mass, in grams, of h2 that must have reacted, to the correct number of significant figures?
0.1
0.102
0.10209
0.1021

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:20
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 14:00
Which of the following is true about a carbonated soft drink? . the carbon dioxide is the solvent, and water is the solute.. the water is the solution, and carbon dioxide is the solvent.. the carbon dioxide is the solution, and the water is the solvent.. the water is the solvent, and the carbon dioxide is the solute.. .
Answers: 1

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 22.06.2019 15:30
The reactions of photosynthesis occur in the of plant cell? a.mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1
You know the right answer?
The haber process can be used to produce ammonia (nh3) from hydrogen gas (h2) and nitrogen gas (n2)....
Questions



English, 10.12.2020 17:40



Social Studies, 10.12.2020 17:40

Mathematics, 10.12.2020 17:40



Mathematics, 10.12.2020 17:40




Mathematics, 10.12.2020 17:40






 in grams is 0.102g.
in grams is 0.102g.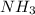 = 17.03 g/mole
= 17.03 g/mole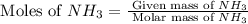 =
= 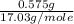 = 0.0337 moles
= 0.0337 moles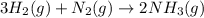
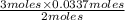 moles of
moles of 

