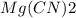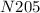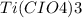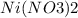1. what is the correct formula for magnesium cyanide?
a. mgcy
b. mncn
c. mg(cn)2...

1. what is the correct formula for magnesium cyanide?
a. mgcy
b. mncn
c. mg(cn)2
d. mgcn
2. what is the correct formula for dinitrogen pentoxide?
a.2no5
b. n2o5
c. dnpo
d. no2.5
3. what is the correct formula for titanium (iii) perchlorate?
a. tipc
b. ti3(clo)4
c. ti3clo4
d. ti(clo4)3
4. what is the correct formula for nickel (iii) nitrate?
a. ni(no3)3
b. ni3no3
c. ni3n
d. nin3
5. what is the correct name for co(c2h3o2)3
a. carbon oxide hydride
b. cobalt acetate
c. cobalt triacetate
d. cobalt (iii) acetate
6. what is the correct name for niso4?
a. nickel (i) sulfide
b. nickel (ii) sulfate
c. nickel sulfide tetraoxide
d. nickel sulfate
7. what is the correct name for n2f4?
a. dinitrogen fluoride ()
b. nitrogen (ii) fluoride
c. dinitrogen tetrafluoride
d. nitrofluorine
8. what is the correct name for pcl5?
a. phosphorus pentachloride
b. phosphorus (v) chloride
c. chlorophosphorus (v)
d. monophosphorus heptachloride
9. which type of bond (ionic, covalent or metallic) would sr and sc make?
a. ionic
b. covalent
c. metallic
d. none of the above
10. which type of bond (ionic, covalent or metallic) would ba and br make?
a. ionic
b. covalent
c. metallic
d. none of the above
11. which type of bond (ionic, covalent or metallic) would copper and tin make?
a. ionic
b. covalent
c. metallic
d. none of the above
12. which type of bond (ionic, covalent or metallic) would n and br make?
a. ionic
b. covalent
c. metallic
d. none of the above

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Identify the electromagnets with poles that are reversed from the electromagnet shown above
Answers: 3

Chemistry, 22.06.2019 12:30
Sodium sulfate dissolves as follows: na2so4(s) → 2na+(aq) + so42- (aq). how many moles of na2so4 are required to make 1.0 l of solution in which the na concentration is 0.10 m?
Answers: 2

Chemistry, 22.06.2019 20:30
Identify the correct mole ratio for each substance. sodium chloride (nacl) na: cl = 1: ammonium nitrate (nhno) h: o = 4:
Answers: 1

Chemistry, 22.06.2019 23:00
How does the value of the equilibrium constant show that a reaction reaches equilibrium very quickly? (a) the equilibrium constant is large. (b) the equilibrium constant is small. (c) the equilibrium constant is zero. (d) the value of the equilibrium constant does not show how quickly a reaction comes to equilibrium.
Answers: 1
You know the right answer?
Questions


Health, 20.07.2019 14:30






Chemistry, 20.07.2019 14:30

Mathematics, 20.07.2019 14:30


History, 20.07.2019 14:30

Business, 20.07.2019 14:30



History, 20.07.2019 14:30



Mathematics, 20.07.2019 14:30

Business, 20.07.2019 14:30