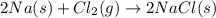Chemistry, 02.01.2020 15:31 jgnjanjdknsjrr9358
Pure sodium metal explodes when it makes contact with water. in its natural state, chlorine is a deadly, poisonous gas. when these two chemicals combine, they form a harmless, white powder known as sodium chloride or table salt. which of the following statements does this information support? a. the properties of substances change during chemical reactions but then later go back to their original condition. b. heat must be added in order to change the chemical properties of substances. c. when substances are chemically combined, the properties of the new substances that are formed are often different from the properties of the original substances. d. when substances are chemically combined, the properties of the new substances that are formed are the same as the properties of the original substances.
i think it's d but i'm not sure

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:30
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 05:50
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2

Chemistry, 22.06.2019 10:20
In a reaction equation, where are the products located? a.) above the arrow b.) to the right of the arrow c.) to the left of the arrow d.) below the arrow
Answers: 2

Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3
You know the right answer?
Pure sodium metal explodes when it makes contact with water. in its natural state, chlorine is a dea...
Questions

Physics, 18.03.2021 02:00

Mathematics, 18.03.2021 02:00

Chemistry, 18.03.2021 02:00

Chemistry, 18.03.2021 02:00

Chemistry, 18.03.2021 02:00


Biology, 18.03.2021 02:00

Law, 18.03.2021 02:00


Biology, 18.03.2021 02:00



History, 18.03.2021 02:00


History, 18.03.2021 02:00





Chemistry, 18.03.2021 02:00