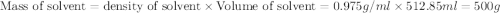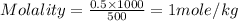Chemistry, 22.12.2019 21:31 amandasantiago2001
Given that the density of water is 0.975 g/ml and that 171 g of sucrose (molar mass: 342.30 g/mol) is dissolved in 512.85 ml of water at 80°c, what is the molality of this solution?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
Which of the following pairs of elements belong to the same groupa. h and he b. li and bec. c and pb d. ga and ge
Answers: 1

Chemistry, 22.06.2019 13:10
Which electron configuration represents the electrons in an atom of sodium in the ground state at stp
Answers: 1

Chemistry, 22.06.2019 21:30
What is happening when the water inside a kettle heats up and begins to boil
Answers: 1

Chemistry, 23.06.2019 04:50
The diagin dilutepage 6 of 12a6a5(a)fluorine, chlorine, bromine and iodine are placed in the same group of theperiodic table.state the common name used to describe elements in this group.(i)state the group in which the elements are placed and explain whythey are placed in that group.(ii)which of the above named elements is a solid at roomtemperature and pressure?
Answers: 2
You know the right answer?
Given that the density of water is 0.975 g/ml and that 171 g of sucrose (molar mass: 342.30 g/mol)...
Questions


Computers and Technology, 25.10.2020 19:30





Mathematics, 25.10.2020 19:30

History, 25.10.2020 19:30

Mathematics, 25.10.2020 19:30

French, 25.10.2020 19:30

History, 25.10.2020 19:30

Mathematics, 25.10.2020 19:30



Computers and Technology, 25.10.2020 19:30

Physics, 25.10.2020 19:30


Mathematics, 25.10.2020 19:30

Chemistry, 25.10.2020 19:30

Biology, 25.10.2020 19:30

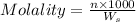
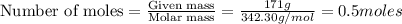
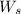 = weight of solvent in g
= weight of solvent in g