

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 23.06.2019 03:50
What is the equation fort the alkaline zinc/manganese dioxide cell. a) anode b)cathode c)overall equations.
Answers: 2

Chemistry, 23.06.2019 08:00
What is the temperature in kelvin of a gas if it is allowed to expand from 1.50 l to 4.50 l? the initial temperature is 10.0°c and pressure is constant throughout the change. which equation should you use? t2= v2/v1 t1 what is the final temperature? ⇒ 849 k these are the answers.
Answers: 1
You know the right answer?
Asealed flask contains 1.3 x 10^25 molecules of co2. how many moles of co2 are in the flask?...
Questions



Mathematics, 09.01.2021 19:20

Mathematics, 09.01.2021 19:20

Computers and Technology, 09.01.2021 19:20



History, 09.01.2021 19:20

Mathematics, 09.01.2021 19:20

Mathematics, 09.01.2021 19:20

History, 09.01.2021 19:20


English, 09.01.2021 19:20





Mathematics, 09.01.2021 19:20


Geography, 09.01.2021 19:20

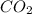 in the flask are, 21.58 moles.
in the flask are, 21.58 moles.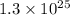 . Now we have to calculate the moles of
. Now we have to calculate the moles of 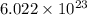 molecules of
molecules of 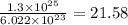 moles of
moles of 

