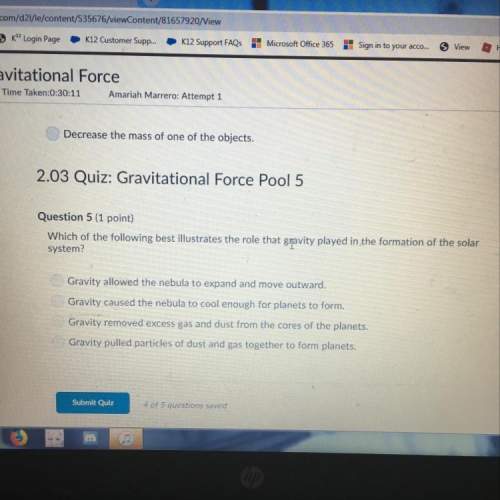
Which reactions show an oxide acting as a base?
check all that apply.
k2o(s)+h2o(...

Chemistry, 02.02.2020 19:57 xdimplesjoon
Which reactions show an oxide acting as a base?
check all that apply.
k2o(s)+h2o(l)→2k+(aq)+oh−(aq)
so3(l)+h2o(l)→ h+(aq)+hso4−(aq)
al2o3(s)+6oh−(aq)+3h2o(l)→2al(oh)−4 (aq)
al2o3(s)+6h+(aq)→2al3+(aq)+3h2o(l)< br /> sio2(s)+2oh−(aq)→ sio3−(aq)+h2o(l)
mgo(s)+2h+(aq)→mg2+(aq)+h2o(l)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 18:50
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3

Chemistry, 22.06.2019 22:30
Which of the following molecules is polar? c3h7oh c2h5cooh
Answers: 1

Chemistry, 23.06.2019 02:30
Calculate the ph at the equivalence point for the titration of a solution containing 150.0 mg of ethylamine (c2h5nh2) with 0.1000 m hcl solution. the volume of the solution at the equivalence point is 250.0 ml. kb forethylamine is 4.7 × 10−4 .
Answers: 2

Chemistry, 23.06.2019 05:30
Based on the formulas, select the compounds below that are covalent: kbr sif4 al2o3 co2 naco3 s7o2 pcl3 fe3n2 h2o s2f10
Answers: 3
You know the right answer?
Questions







Mathematics, 18.01.2021 14:00


Computers and Technology, 18.01.2021 14:00

Social Studies, 18.01.2021 14:00

Mathematics, 18.01.2021 14:00



Computers and Technology, 18.01.2021 14:00

Mathematics, 18.01.2021 14:00


Mathematics, 18.01.2021 14:00


English, 18.01.2021 14:00