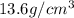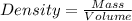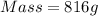Chemistry, 14.11.2019 03:31 JadeCaldwell
The density of mercury is 13.6 g/cm3. what approximate mass of mercury is required to fill a 2.0 ounce bottle? an ounce is approximately 30 cm3.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:30
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1


Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1
You know the right answer?
The density of mercury is 13.6 g/cm3. what approximate mass of mercury is required to fill a 2.0 oun...
Questions




Spanish, 03.12.2020 07:20

Advanced Placement (AP), 03.12.2020 07:20

English, 03.12.2020 07:20


Mathematics, 03.12.2020 07:20

Mathematics, 03.12.2020 07:20



English, 03.12.2020 07:20

Mathematics, 03.12.2020 07:20


Social Studies, 03.12.2020 07:20

Mathematics, 03.12.2020 07:20

Biology, 03.12.2020 07:20

English, 03.12.2020 07:20


Chemistry, 03.12.2020 07:20