
Chemistry, 22.09.2019 14:00 StupidFatChipmunk
If you had excess chlorine, how many moles of of aluminum chloride could be produced from 19.0 g of aluminum?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3

Chemistry, 22.06.2019 20:30
How many grams of phosphorus are contained in 5.09 moles of phosphorus?
Answers: 1

Chemistry, 22.06.2019 22:30
Which is a characteristic of the electron sea model for metallic bonding? molecular orbitals overlap to produce bands. electrons flow easily between metal nuclei. electrons are in fixed positions in the orbitals. atomic nuclei are arranged in an irregular pattern.
Answers: 3

Chemistry, 23.06.2019 02:00
Pinene is an unsaturated hydrocarbon found in pine resin. if pinene has m+ = 136 and contains 1 double bond(s) and 2 ring(s); what is its molecular formula? enter the formula in the form ch first, then all other atoms in alphabetical order; do not use subscripts. the formula is case-sensitive
Answers: 3
You know the right answer?
If you had excess chlorine, how many moles of of aluminum chloride could be produced from 19.0 g of...
Questions

History, 18.10.2020 08:01

Mathematics, 18.10.2020 08:01


French, 18.10.2020 08:01

Chemistry, 18.10.2020 08:01

English, 18.10.2020 08:01

Mathematics, 18.10.2020 08:01


History, 18.10.2020 08:01




Mathematics, 18.10.2020 08:01

Mathematics, 18.10.2020 08:01




Mathematics, 18.10.2020 08:01


English, 18.10.2020 08:01

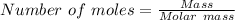
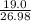
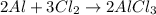
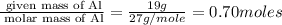
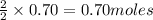 of aluminium chloride.
of aluminium chloride.

