
Chemistry, 31.01.2020 01:59 keananashville
What are the resulting coefficients when you balance the chemical equation for the combustion of ethane, c2h6? in this reaction, ethane is burned in the presence of oxygen (o2) to form carbon dioxide (co2) and water (h2o). (g)+(g)→(g)+(g)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Asample of the male sex hormone testosterone, c19h28o2, contains 3.88×10^21 atoms of hydrogen.(a) how many atoms of carbon does it contain? (b) how many molecules of testosterone does it contain? (c) how many moles of testosterone does it contain? (d) what is the mass of this sample in grams?
Answers: 1


Chemistry, 23.06.2019 05:30
Find the midpoint of a segment with endpoints of 4-3i and -2+7i
Answers: 2

Chemistry, 23.06.2019 15:30
How many milliliters of 0.550 m hydriodic acid are needed to react with 25.00 ml of 0.217 m csoh?
Answers: 2
You know the right answer?
What are the resulting coefficients when you balance the chemical equation for the combustion of eth...
Questions

Biology, 27.09.2021 01:00


Mathematics, 27.09.2021 01:00

Computers and Technology, 27.09.2021 01:00

Mathematics, 27.09.2021 01:00


History, 27.09.2021 01:00

Biology, 27.09.2021 01:00

Mathematics, 27.09.2021 01:00

Spanish, 27.09.2021 01:00



English, 27.09.2021 01:10


Mathematics, 27.09.2021 01:10




English, 27.09.2021 01:10

Biology, 27.09.2021 01:10

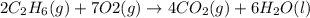
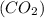 and water
and water 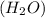 .
.

