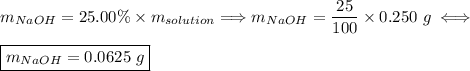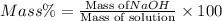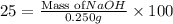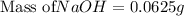Chemistry, 27.09.2019 19:40 samirahscott
Asolution of naoh has a concentration of 25.00% by mass. what mass of naoh is present in 0.250 g of this solution? use the periodic table in the toolbar if needed.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:30
219 grams of iron (iii) oxide reacts with excess carbon according to the reaction equation shown below. fe2o3 + c → fe + co2 after a scientist performs the chemical reaction they find the actual yield of iron to be 57.4 grams. calculate the percent yield of this chemical reaction.
Answers: 1

Chemistry, 21.06.2019 22:20
Which of the following statements is false regarding aromaticity? a. the compound must be cyclic b. the compound must be fully conjugated c. the compound must be planar d.the number of electrons in the pi system must satisfy the hückel 4n+2 rule e. the compound must have a neutral charge
Answers: 2

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

You know the right answer?
Asolution of naoh has a concentration of 25.00% by mass. what mass of naoh is present in 0.250 g of...
Questions

Geography, 05.02.2021 01:00


Computers and Technology, 05.02.2021 01:00

Mathematics, 05.02.2021 01:00

History, 05.02.2021 01:00



Mathematics, 05.02.2021 01:00

Social Studies, 05.02.2021 01:00

Physics, 05.02.2021 01:00

Computers and Technology, 05.02.2021 01:00


Mathematics, 05.02.2021 01:00

Mathematics, 05.02.2021 01:00



Mathematics, 05.02.2021 01:00



Arts, 05.02.2021 01:00