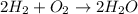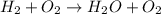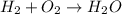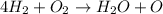Chemistry, 20.10.2019 06:50 samanthahurtado0914
Which of the following equations is balanced?
h2 + o2→h2o + o2
2h2 + o2→2h2o
h2+ o2→h2o
4h2 + o2→h2o + o

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:30
When svante arrhenius first proposed his acid-base theory, he was a doctoral candidate. his professors thought his ideas were unfounded. within a decade, the arrhenius theory of acid-base was widely accepted and praised within the scientific world. arrhenius defined acids as compounds having ionizable hydrogen and bases as compounds with ionizable a) barium. b) hydronium. c) hydroxide. d) oxygen.
Answers: 3

Chemistry, 22.06.2019 13:00
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1

Chemistry, 22.06.2019 16:00
1. an experiment in your science class lists the materials needed for the lab. it is your job, as a lab partner, to measure out 25 ml of distilled water and 2.5 grams of magnesium. what lab measuring tools would you choose to measure each substance and how would you use each tool to get the correct amounts? be sure to describe the process you would follow step-by-step. (5 points) 2.which of the following is an si base unit for measuring mass? (2 points) ampere gram meter pound 3.which of the following is an si base unit for time? (2 points) decades hours minutes seconds 4.which of the following tools should a scientist use to measure an object in milligrams? (2 points) graduated cylinder pan balance tape measure thermometer 4.which of the following tools should a scientist use to measure an object in milligrams? (2 points) graduated cylinder pan balance tape measure thermometer. 5.a pencil beside a metric ruler. the ruler is scaled from 1 centimeter to 10 centimeters, with markings for millimeters between each number. one end of the pencil is beside the 0 on the ruler, and the pencil point is beside the 5. which of the following measurements is accurate but not precise? (2 points) 5 mm 5 cm 50 mm 50 cm 6. which of the following prefixes represents the largest value? (2 points) giga hector kilo milli 7. which of the following types of graphs is best for plotting the percentages of a whole value in a data set? (2 points) bar graph circle graph histogram line graph
Answers: 1

Chemistry, 23.06.2019 06:00
•what conclusions can you make about the relationship between the volume of a gas and its temperature? • what conclusions can you make about the relationship between the volume of a gas and its pressure? • what possible variables have you not accounted for? as you did the procedures, is it possible that the atmospheric pressure may have changed? if it did change over the course of your experiment, then how would your results have been affected?
Answers: 3
You know the right answer?
Which of the following equations is balanced?
h2 + o2→h2o + o2
2h2 + o2→2h2...
h2 + o2→h2o + o2
2h2 + o2→2h2...
Questions

Biology, 18.11.2020 09:00


Computers and Technology, 18.11.2020 09:00



Mathematics, 18.11.2020 09:00


Mathematics, 18.11.2020 09:00

Mathematics, 18.11.2020 09:00

Mathematics, 18.11.2020 09:00

Mathematics, 18.11.2020 09:00


Physics, 18.11.2020 09:00

Physics, 18.11.2020 09:00

English, 18.11.2020 09:00


Mathematics, 18.11.2020 09:00

History, 18.11.2020 09:00