
Chemistry, 03.10.2019 12:50 andreastyles1603
Measurements show that enthalpy of a mixture of gaseous reactants decreases by 228. kj during a certain chemical reaction, which is carried out at a constant pressure. furthermore, by carefully monitoring the volume change it is determined that -55kj of work is done on the mixture during the reaction.
calculate the change in energy of the gas mixture during the reaction.
is the reaction exothermic or endothermic?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1

Chemistry, 22.06.2019 07:20
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1

Chemistry, 23.06.2019 07:30
Which statement explains which thermometer is more appropriate to measure the temperature of a liquid at 43.6 degrees celsius a) thermometer a, because it measures temperature more accurately than thermometer b b) thermometer b, because it measures temperature more accurately than thermometer a c) thermometer a, because it measures temperature more precisely than thermometer b d) thermometer b, because it measures temperature more precisely than thermometer a
Answers: 2

Chemistry, 23.06.2019 07:40
Which of the following has expanded our knowledge of the universe beyond our solar system the most? a. manned space travel b. the hubble space telescope c. the pioneer and voyager missions d. the international space station
Answers: 3
You know the right answer?
Measurements show that enthalpy of a mixture of gaseous reactants decreases by 228. kj during a cert...
Questions



Mathematics, 18.03.2021 01:20

Physics, 18.03.2021 01:20


Mathematics, 18.03.2021 01:20

Computers and Technology, 18.03.2021 01:20



Computers and Technology, 18.03.2021 01:20

History, 18.03.2021 01:20


Geography, 18.03.2021 01:20





Mathematics, 18.03.2021 01:20


Social Studies, 18.03.2021 01:20

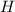 =-228 KJ
=-228 KJ 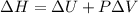
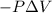
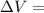 Change in volume
Change in volume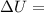 Change in energy
Change in energy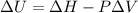

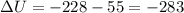 KJ
KJ

