
Chemistry, 19.04.2021 18:00 muncyemily
Calculate the number of water (H2O) molecules produced from the decomposition of 75.50 grams of Iron (III) hydroxide (Fe(OH)3).

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 21:00
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1

Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 05:40
Convert a speed of 201 cm/s to units of inches per minute. also, show the unit analysis by dragging components into the unit‑factor slots.
Answers: 1

Chemistry, 23.06.2019 12:00
Which element has the largest atomic radius? a. asb. nc. pd. sb
Answers: 2
You know the right answer?
Calculate the number of water (H2O) molecules produced from the decomposition of 75.50 grams of Iron...
Questions

Mathematics, 11.09.2020 14:01

English, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

English, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Social Studies, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

English, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

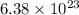 molecules of water are produced.
molecules of water are produced.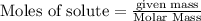
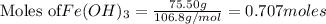
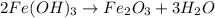
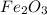 produce = 3 moles of
produce = 3 moles of 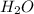
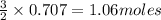 of
of 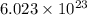 of particles.
of particles.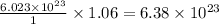 molecules
molecules

