Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1


Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2

Chemistry, 23.06.2019 03:30
Calculate the ph of a .10m nh4cl solution. the kb value for nh3 is 1.8×10^-5
Answers: 1
You know the right answer?
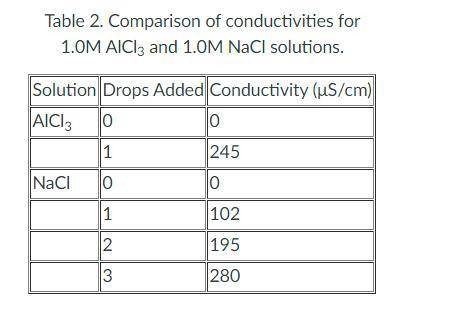
How to find molar conductivity (in µS/M·cm) given 1.0M and a table of conductivity?
I think I have...
Questions

English, 15.09.2021 19:30



Biology, 15.09.2021 19:30




Mathematics, 15.09.2021 19:30

History, 15.09.2021 19:30


Chemistry, 15.09.2021 19:30



Mathematics, 15.09.2021 19:30




Mathematics, 15.09.2021 19:30

Chemistry, 15.09.2021 19:30

English, 15.09.2021 19:30