S8 +O2 = SO8
Given 80 mols of product, find the amount of each reactant in mols....

Chemistry, 19.04.2021 06:00 AdoreeLayla
S8 +O2 = SO8
Given 80 mols of product, find the amount of each reactant in mols.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 21.06.2019 23:00
The wave shown on the electromagnetic spectrum disturb the medium it passes through a)different frequency. b)the same frequency .
Answers: 2

Chemistry, 22.06.2019 05:50
According to coulomb's law, how would the electrical force between particles change if the product of their electrical charge increased?
Answers: 1

Chemistry, 22.06.2019 08:30
In the reaction between a crushed antacid tablet and vinegar what gas is emitted
Answers: 2
You know the right answer?
Questions

English, 04.03.2021 22:00

Mathematics, 04.03.2021 22:00

Mathematics, 04.03.2021 22:00



Mathematics, 04.03.2021 22:00


Chemistry, 04.03.2021 22:00

Health, 04.03.2021 22:00

English, 04.03.2021 22:00


Mathematics, 04.03.2021 22:00

Mathematics, 04.03.2021 22:00

English, 04.03.2021 22:00




Mathematics, 04.03.2021 22:00


Mathematics, 04.03.2021 22:00

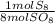 = 10 mol S₈80 mol SO₈ *
= 10 mol S₈80 mol SO₈ * 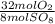 = 320 mol O₂
= 320 mol O₂

