
Chemistry, 16.04.2021 20:40 homeschool0123
An analytical chemist is titrating 111.0 mL of a 0.3700 M solution of aniline (C6H5NH2) with a 0.3500 M solution of HNO3. The pK_b of aniline is 9.37. Calculate the pH of the base solution after the chemist has added 79.1 mL of the HNO_3 solution to it.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 20:30
Select all the correct answers.which compounds have the empirical formula ch20? (multiple answers)a.c2h4o2b.c3h603c.ch2o2d.c5h1005e.c6h1206
Answers: 2

Chemistry, 22.06.2019 23:30
Substance a is a nonpolar liquid and has only dispersion forces among its constituent particles. substance b is also a nonpolar liquid and has about the same magnitude of dispersion forces among its constituent particles. when substance a and b are combined, they spontaneously mix.
Answers: 1

Chemistry, 23.06.2019 10:10
In a covalent bond, two atoms are held together by the attraction between . the number of covalent bonds that an atom can form depends on the number of in the atom.
Answers: 2
You know the right answer?
An analytical chemist is titrating 111.0 mL of a 0.3700 M solution of aniline (C6H5NH2) with a 0.350...
Questions



Mathematics, 02.03.2020 04:51

Biology, 02.03.2020 04:51



Physics, 02.03.2020 04:52


Geography, 02.03.2020 04:52


English, 02.03.2020 04:52


Chemistry, 02.03.2020 04:52

Computers and Technology, 02.03.2020 04:52



Social Studies, 02.03.2020 04:54

Biology, 02.03.2020 04:54


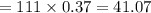
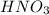 millimoles
millimoles 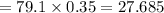
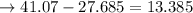 millimoles aniline left
millimoles aniline left
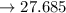 millimoles salt formed
millimoles salt formed
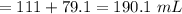
![\to [aniline] = \frac{13.385}{190.1} = 0.07 \ M\\\\\to [salt] =\frac{ 27.685}{ 190.1} = 0.146\ M\\\\\to pOH = pKb + \frac{\log [salt]}{ [base]}\\\\\to pOH = 9.37 + \frac{\log [0.146]}{[0.07]}\\\\\to pOH = 9.69\\\\\to pH = 14 - 9.69\\\\\to pH = 4.31\\](/tpl/images/1264/8201/7a097.png)


