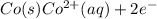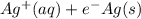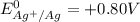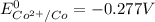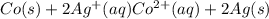Chemistry, 16.04.2021 18:00 Simplytaylorgrenade
The half-cell is a chamber in the voltaic cell where one half-cell is the site of the oxidation reaction and the other half-cell is the site of the reduction reaction. Type the half-cell reaction that takes place at the anode for the cobalt-silver voltaic cell. Indicate the physical states using the abbreviation (s), (l), or (g) for solid, liquid, or gas, respectively. Use (aq) for an aqueous solution. Do not forget to add electrons in your reaction.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Plz choose one of the compounds from the table and explain how you know the numbers of atoms in your formula. is it possible for two different compounds to be made from the exact same two elements? why or why not? with a limited number of elements (less than 120 are known), does this mean we also have a small number of compounds or do we have a large number of compounds in this world?
Answers: 1


Chemistry, 22.06.2019 16:30
4. a 20-kg child is tossed up into the air by her parent. the child is 2 meters off the ground traveling 5 m/s. circle one: ke / gpe / both show your work for finding the values of each type of energy the object has:
Answers: 1

Chemistry, 22.06.2019 17:00
Reduction is a reaction which results in a in electrons and a in positive charge of the atom or ion 1) a- loss 1) b- gain 2) a-increase 2) b-decrease
Answers: 1
You know the right answer?
The half-cell is a chamber in the voltaic cell where one half-cell is the site of the oxidation reac...
Questions

Mathematics, 17.05.2021 21:30

Mathematics, 17.05.2021 21:30


Mathematics, 17.05.2021 21:30

Social Studies, 17.05.2021 21:30



Arts, 17.05.2021 21:30

English, 17.05.2021 21:30


Mathematics, 17.05.2021 21:30

Mathematics, 17.05.2021 21:30

Mathematics, 17.05.2021 21:30



English, 17.05.2021 21:30

Mathematics, 17.05.2021 21:30


Mathematics, 17.05.2021 21:30