CaCO3 + 2 HI —
Cal2 + H2O + CO2
a) If 4.35 g of CaCOreact with 55.0 mL of 0.25 M HI determine...

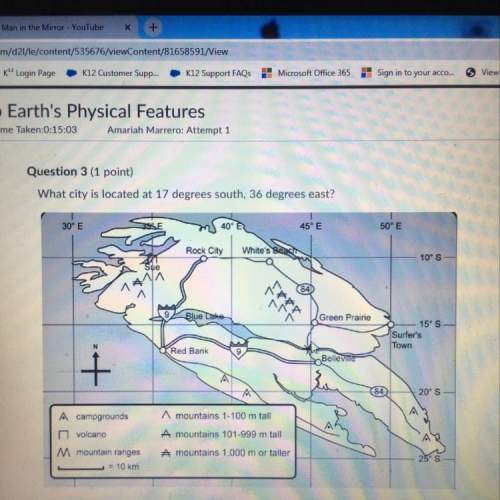
Chemistry, 16.04.2021 15:10 princeofpowerjr
CaCO3 + 2 HI —
Cal2 + H2O + CO2
a) If 4.35 g of CaCOreact with 55.0 mL of 0.25 M HI determine:
8
(i) The volume of CO, that would be formed at STP (theoretical yield)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:40
Which of the following pressures is equal to 760 mm hg? 2.0 atm 101.3 pa 101,300 kpa 101,300 pa
Answers: 2

Chemistry, 22.06.2019 07:00
Which atom or ion is the largest? a. k b. k+ c. ca d. ca2+ e. li
Answers: 1

Chemistry, 22.06.2019 09:00
Suppose you have designed a new thermometer called the x thermometer. on the x scale the boiling point of water is 129 ? x and the freezing point of water is 13 ? x. part a at what temperature are the readings on the fahrenheit and x thermometers the same?
Answers: 1

You know the right answer?
Questions


Mathematics, 18.07.2019 01:30




History, 18.07.2019 01:30

Biology, 18.07.2019 01:30

Mathematics, 18.07.2019 01:30


Physics, 18.07.2019 01:30

Mathematics, 18.07.2019 01:30



Computers and Technology, 18.07.2019 01:30

History, 18.07.2019 01:30


Mathematics, 18.07.2019 01:30

Social Studies, 18.07.2019 01:30