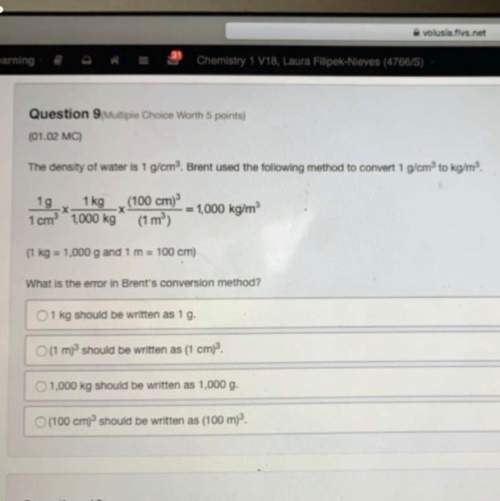
10.0g of solid sodium carbonate and excess of aqueous acetic acid react to produce sodium acetate, carbon dioxide, and water. When the reaction is complete, 1.98 L of CO2 at STP conditions were produced.
Na2CO3(s) +
2 HC2H3O2(aq) à
2 NaC2H3O2(aq) + CO2(g) + H2O(l)
a) Calculate the maximum amount of carbon dioxide that could had been made under these conditions.
b) Calculate the % yield for the reaction.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 19:10
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2


Chemistry, 23.06.2019 10:30
Which of the following characteristics are true of enzymes? check all that apply. a.)the structure of an enzyme can change if conditions change. b.)a single enzyme can normally catalyze a wide variety of reactions under many conditions. c.)enzymes are found only in nonliving systems. d.)enzymes allow living things to regulate body conditions through feedback mechanisms. e.)enzymes bind to specific substrates in specific ways. f.)enzymes increase the rate of reaction. g.)when shown in energy-reaction diagrams, enzymes represent the higher activation energy.
Answers: 1

You know the right answer?
10.0g of solid sodium carbonate and excess of aqueous acetic acid react to produce sodium acetate, c...
Questions



Health, 03.02.2020 12:47






Mathematics, 03.02.2020 12:47


Physics, 03.02.2020 12:47


English, 03.02.2020 12:47

Biology, 03.02.2020 12:47



Physics, 03.02.2020 12:47


Social Studies, 03.02.2020 12:47

Mathematics, 03.02.2020 12:47