
Chemistry, 15.04.2021 18:00 rodneydean4515
Drag and drop the labels provided into the boxes below to explain how the Earth-Sun-Moon system results in these seasons on Earth.
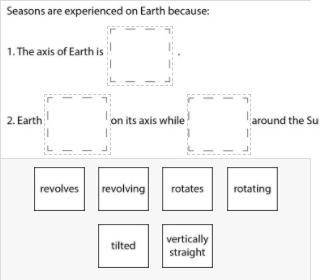

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:40
Determine the energy released per kilogram of fuel used. given mev per reaction, calculate energy in joules per kilogram of reactants. consider 1 mole of tritium plus 1 mole of deuterium to be a mole of “reactions” (total molar mass = 5 grams).
Answers: 1

Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3
You know the right answer?
Drag and drop the labels provided into the boxes below to explain how the Earth-Sun-Moon system resu...
Questions


Mathematics, 07.07.2019 11:30

Biology, 07.07.2019 11:30

Geography, 07.07.2019 11:30


Biology, 07.07.2019 11:30

Mathematics, 07.07.2019 11:30


History, 07.07.2019 11:30

Mathematics, 07.07.2019 11:30

Mathematics, 07.07.2019 11:30

History, 07.07.2019 11:30

Mathematics, 07.07.2019 11:30

Biology, 07.07.2019 11:30


Geography, 07.07.2019 11:30







