Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
219 grams of iron (iii) oxide reacts with excess carbon according to the reaction equation shown below. fe2o3 + c → fe + co2 after a scientist performs the chemical reaction they find the actual yield of iron to be 57.4 grams. calculate the percent yield of this chemical reaction.
Answers: 1

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1


Chemistry, 23.06.2019 02:50
Dumbledore decides to gives a surprise demonstration. he starts with a hydrate of na2co3 which has a mass of 4.31 g before heating. after he heats it he finds the mass of the anhydrous compound is found to be 3.22 g. he asks everyone in class to determine the integer x in the hydrate: na2co3·xh2o; you should do this also. round your answer to the nearest integ
Answers: 2
You know the right answer?
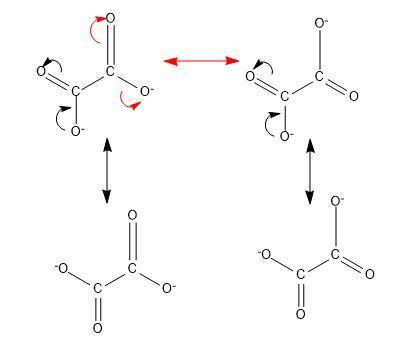
Determine the number of resonance structures for the oxalate ion, c2o42−.
2
...
2
...
Questions

Social Studies, 17.10.2020 15:01



English, 17.10.2020 15:01



English, 17.10.2020 15:01


Social Studies, 17.10.2020 15:01


Mathematics, 17.10.2020 15:01


History, 17.10.2020 15:01


English, 17.10.2020 15:01

Chemistry, 17.10.2020 15:01

English, 17.10.2020 15:01


Mathematics, 17.10.2020 15:01

Mathematics, 17.10.2020 15:01