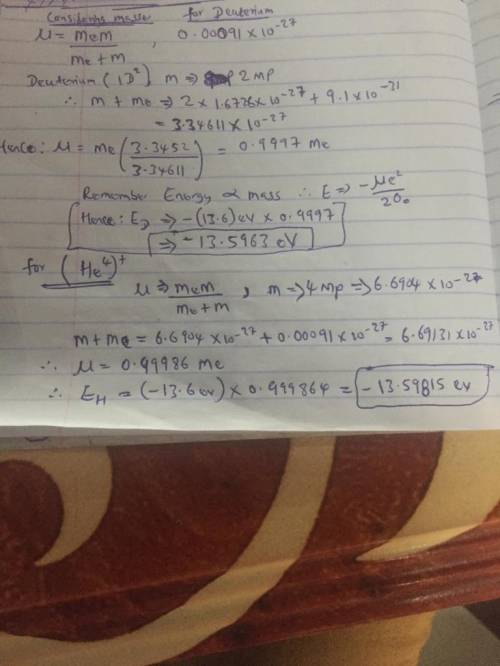Chemistry, 13.04.2021 03:00 kamnicole13
Hydrogen, deuterium, and singly ionized helium are all examples of one-electronatoms. The deuterium nucleus has the same charge as the hydrogen nucleus, and almost exactlytwice the mass. The helium nucleus has twice the charge of the hydrogen nucleus, and almostexactly four times the mass. Make a prediction of the ratios of the ground state energies of theseatoms, considering that current spectroscopy accuracy is extremely good (on the order of107).

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:30
The mass of a neutron is equal to the mass of a proton plus the mass of an electron. true or false false true
Answers: 1

Chemistry, 22.06.2019 16:00
Which factor is likely to impact the possible number of compounds ?
Answers: 1

Chemistry, 22.06.2019 23:10
Amines are good nucleophiles, even though they are neutral molecules. how would the rate of an sn2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? amines are good nucleophiles, even though they are neutral molecules. how would the rate of an reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? because both reactants in the rate-limiting step are neutral, the reaction will be faster if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will be slower if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will occur at the same rate if the polarity of the solvent is increased. request answer
Answers: 3

You know the right answer?
Hydrogen, deuterium, and singly ionized helium are all examples of one-electronatoms. The deuterium...
Questions

Mathematics, 11.11.2020 19:40

Engineering, 11.11.2020 19:50

Mathematics, 11.11.2020 19:50





Mathematics, 11.11.2020 19:50



Mathematics, 11.11.2020 19:50

Chemistry, 11.11.2020 19:50

Mathematics, 11.11.2020 19:50

Computers and Technology, 11.11.2020 19:50




Mathematics, 11.11.2020 19:50