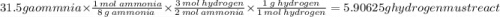Chemistry, 12.04.2021 17:40 reneethacker20p8wdgn
Hydrogen and nitrogen react to produce ammonia gas as shown in the following chemical
equation.
N2 + 3H2 - 2NH:
How many grams of hydrogen must react to produce 31.5 grams of ammonia?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:50
Ase your answer to this question on the information below.hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant.the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen.chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product.nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1

Chemistry, 22.06.2019 04:30
There is a single path for electrons. the current decreases when additional resistors are added. the current will be the same in each resistor. these statements best describe a(n) circuit.
Answers: 3

Chemistry, 22.06.2019 05:00
When you mate two plants together the terms is called? answer it fast as possible plz! i have a test tomorrow!
Answers: 1

Chemistry, 23.06.2019 01:00
The primary products of complete combustion of fossil fuels are a. carbon dioxide and water b. methane and water c. carbon monoxide and water d. carbon dioxide and carbon monoxide
Answers: 1
You know the right answer?
Hydrogen and nitrogen react to produce ammonia gas as shown in the following chemical
equation.
Questions

Mathematics, 06.10.2020 14:01

Advanced Placement (AP), 06.10.2020 14:01

Mathematics, 06.10.2020 14:01


Mathematics, 06.10.2020 14:01


Mathematics, 06.10.2020 14:01


History, 06.10.2020 14:01


Biology, 06.10.2020 14:01


Mathematics, 06.10.2020 14:01

Biology, 06.10.2020 14:01



History, 06.10.2020 14:01