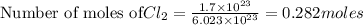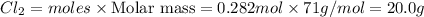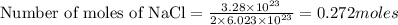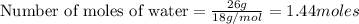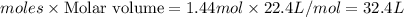1. How many grams are in 1.7 x 10^23 particles of Cl2?
2. How many moles are in 3.28 x 10^23 atoms of NaCl? *
3. If I were to determine how many liters 26 grams of water is, what type of conversion would this be? *
A Mass --> Moles --> Particles
B Mass --> Moles --> Volume
C Volume --> Mass --> Moles
D Moles --> Mass --> Volume

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 15:00
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

Chemistry, 22.06.2019 19:00
Sum of brother and sisters age is 26. four times the brothers age is subtracted from three times the sisters age, the difference is 8. what are the ages of the brother and sister?
Answers: 1

Chemistry, 22.06.2019 21:20
40dm3 of gas at 760 torr are heated from 5°c to 50°c what is the new volume
Answers: 3

Chemistry, 22.06.2019 22:00
Plz ill give u brainliest which of the following steps is not likely to take place during cellular respiration? a.oxygen combines with carbon of simple sugar. b. energy molecule transfers energy to cells. d. energy is used up.
Answers: 3
You know the right answer?
1. How many grams are in 1.7 x 10^23 particles of Cl2?
2. How many moles are in 3.28 x 10^23 atoms...
Questions


English, 13.07.2019 01:30









Mathematics, 13.07.2019 01:30






Mathematics, 13.07.2019 01:30



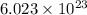 of particles.
of particles.
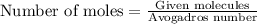 or
or 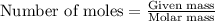 or
or