Will give brainliest!!
Iodine (V) oxide (I2O5) and carbon monoxide (CO) react to produce carbon dioxide and iodine according to the equation: I2O5 + 5CO -> 5CO2 + I2
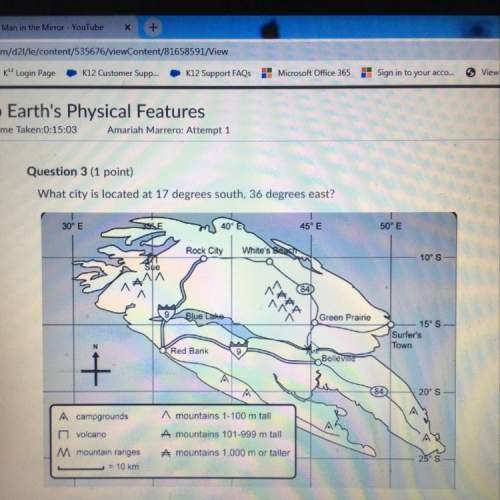
How many moles of Iodine (V) oxide (I2O5) are needed to react with 3.50 moles of carbon monoxide (CO)?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Will mark brainliest 26. which of these statements are true? (3 points) a. gases are compressible b. gases fill their containers completely c. the pressure of a gas is independent of the temperature d. gases have mass e. gases exert pressure f. the pressure of a gas is dependent on the volume g. gas pressure results from the collisions between gas particles h. gases have a definite volume and shape
Answers: 1


Chemistry, 22.06.2019 04:10
Answer from each drop-down menu. e characteristics of a borane molecule (bh). the lewis structure and table of electronegativities are given olecular shape is and the molecule is reset next erved. search e a
Answers: 2

Chemistry, 22.06.2019 10:30
Which characteristics can be used to differentiate star systems? check all that apply.
Answers: 2
You know the right answer?
Will give brainliest!!
Iodine (V) oxide (I2O5) and carbon monoxide (CO) react to produce carbon dio...
Questions


Chemistry, 18.07.2019 17:00





Mathematics, 18.07.2019 17:00

Mathematics, 18.07.2019 17:00

History, 18.07.2019 17:00







Computers and Technology, 18.07.2019 17:00


Computers and Technology, 18.07.2019 17:00

Mathematics, 18.07.2019 17:00