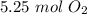Consider the following balanced equation:
2KCIO3(s) → 2KCl(s) + 302(g)
How many moles of O2 w...

Chemistry, 06.04.2021 23:20 hncriciacheichi
Consider the following balanced equation:
2KCIO3(s) → 2KCl(s) + 302(g)
How many moles of O2 will be obtained by decomposing
3.50 moles of KCIO3?
0.530 mole
O 3.00 moles
O 2.30 moles
5.25 moles

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Apiece of metal with a length of 1.42 cm was measured using four different instruments. which of the following measurements is the most accurate?
Answers: 3

Chemistry, 22.06.2019 18:30
Which sample at stp has the same number of atoms as 18 liters of ne at stp
Answers: 1

Chemistry, 22.06.2019 18:40
What is the binding energy of a nucleus that has a mass defect of 5.81*10-^29 kg a 5.23*10-^12 j b 3.15* 10^12 j c 1.57*10-3 j d 9.44*10^20 j
Answers: 1

Chemistry, 22.06.2019 20:30
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1
You know the right answer?
Questions

Business, 03.11.2020 16:00



Mathematics, 03.11.2020 16:00

Mathematics, 03.11.2020 16:00



Chemistry, 03.11.2020 16:00



English, 03.11.2020 16:00

Computers and Technology, 03.11.2020 16:00

English, 03.11.2020 16:00

English, 03.11.2020 16:00

Mathematics, 03.11.2020 16:00


Mathematics, 03.11.2020 16:00


Mathematics, 03.11.2020 16:00

History, 03.11.2020 16:00

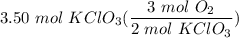 [DA} Multiply [Cancel out units]:
[DA} Multiply [Cancel out units]: