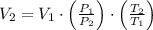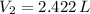Chemistry, 02.04.2021 16:40 rcmhargoux2970
A child has a toy balloon with a volume of 1.80 liters. The temperature of the balloon when it was filled was 20° C and the pressure was 1.00 atm. If the child were to let go of the balloon and it rose 3 kilometers into the sky where the pressure is 0.667 atm and the temperature is -10° C, what would the new volume of the balloon be? (Don't forget to convert the temperature to K)

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 20:00
I’m an electrically neutral atomic any element, there are equal numbers of
Answers: 2

Chemistry, 22.06.2019 22:30
Is the idea of spontaneous generation supported by redi's experiment? justify your answer in 2-3 sentences?
Answers: 1
You know the right answer?
A child has a toy balloon with a volume of 1.80 liters. The temperature of the balloon when it was f...
Questions


Mathematics, 03.10.2020 01:01

Computers and Technology, 03.10.2020 01:01

Mathematics, 03.10.2020 01:01

Computers and Technology, 03.10.2020 01:01


Mathematics, 03.10.2020 01:01

Chemistry, 03.10.2020 01:01

Health, 03.10.2020 01:01

Mathematics, 03.10.2020 01:01

Business, 03.10.2020 01:01

Health, 03.10.2020 01:01






Mathematics, 03.10.2020 01:01


 ), in atmospheres, is inversely proportional to volume (
), in atmospheres, is inversely proportional to volume ( ), in liters, and directly proportional to temperature (
), in liters, and directly proportional to temperature (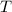 ), in Kelvin. Based on this fact, we construct the following relationship:
), in Kelvin. Based on this fact, we construct the following relationship: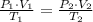 (1)
(1)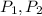 - Initial and final pressures, in atmospheres.
- Initial and final pressures, in atmospheres.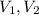 - Initial and final volumes, in liters.
- Initial and final volumes, in liters.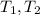 - Initial and final temperatures, in Kelvin.
- Initial and final temperatures, in Kelvin.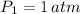 ,
, 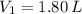 ,
, 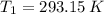 ,
, 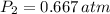 ,
, 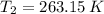 , then the final pressure is:
, then the final pressure is: