
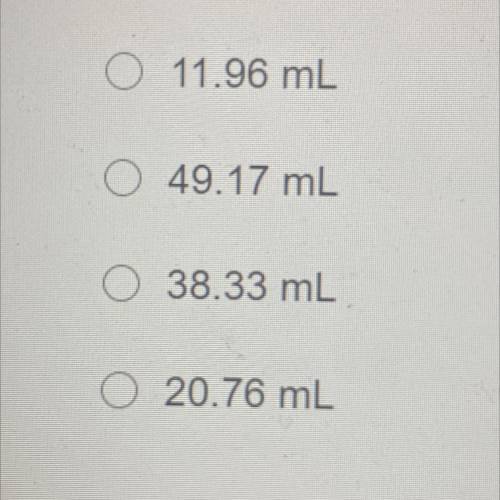
An unopened soda bottle contains 46.0mL of gas confined at a pressure of 1.30 atm at a temperature of 5.0°C. If the bottle is
dropped into a lake and sinks to a depth at which the pressure is 2.85 atm and temperature is 2.09°C, what will the volume of the
gas in the bottle be?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
18. use the activity series to predict whether the following synthesis reaction will occur. write the chemical equations for the reaction if it's predicted to occur. (s) + o2(g) -> *note: it is possible.*
Answers: 1

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 23.06.2019 01:00
The primary products of complete combustion of fossil fuels are a. carbon dioxide and water b. methane and water c. carbon monoxide and water d. carbon dioxide and carbon monoxide
Answers: 1

You know the right answer?
An unopened soda bottle contains 46.0mL of gas confined at a pressure of 1.30 atm at a temperature o...
Questions

Mathematics, 11.02.2021 17:00

Mathematics, 11.02.2021 17:00

Mathematics, 11.02.2021 17:00

Mathematics, 11.02.2021 17:00

Mathematics, 11.02.2021 17:00

Social Studies, 11.02.2021 17:00

Mathematics, 11.02.2021 17:00


Mathematics, 11.02.2021 17:00

Chemistry, 11.02.2021 17:00

Mathematics, 11.02.2021 17:00

Mathematics, 11.02.2021 17:00


Chemistry, 11.02.2021 17:00

History, 11.02.2021 17:00


Mathematics, 11.02.2021 17:00

Mathematics, 11.02.2021 17:00

Biology, 11.02.2021 17:00

Mathematics, 11.02.2021 17:00



