
Chemistry, 01.04.2021 18:40 leonarddyer4599
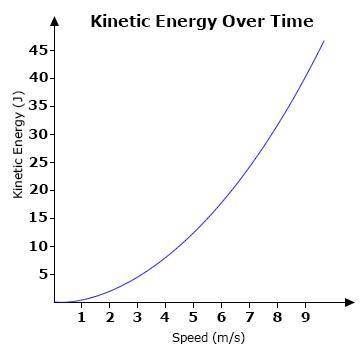
A graph showing the kinetic energy of a ball over time is shown below.
Which of the following statements best explains the relationship between the kinetic energy of the ball and its speed?
A.
The kinetic energy of the ball decreases as its speed increases.
B.
The kinetic energy of the ball remains the same as its speed increases.
C.
The kinetic energy of the ball increases as its speed increases.
D.
The kinetic energy of the ball increases over time as the ball travels at a constant speed.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:30
How many atoms are in 1.4 mil of phosphorus trifluoride (pf3)
Answers: 3


Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 22.06.2019 21:00
One similarity and one difference between an element and a mixture of elements
Answers: 1
You know the right answer?
A graph showing the kinetic energy of a ball over time is shown below.
Which of the following state...
Questions







Mathematics, 14.02.2020 06:20






Mathematics, 14.02.2020 06:21




Mathematics, 14.02.2020 06:24

Mathematics, 14.02.2020 06:24

Social Studies, 14.02.2020 06:28

History, 14.02.2020 06:31



