
Chemistry, 01.04.2021 16:10 Theresab2021
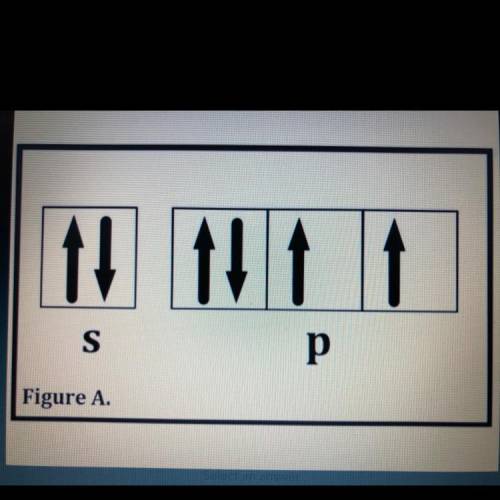
Which of the following elements will have its valence electrons represented as in Figure A?
I. Oxygen and Sulfer
II. Selenium and Tellurium
III. Polonium only
A. I only
B. II only
C. III only
D. I, II, and III

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
In which phase(s) do the molecules take the shape of the container?
Answers: 1

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

Chemistry, 22.06.2019 09:00
The nuclear fission process releases neutrons and question 27 options: alpha particles electrons energy beta particles
Answers: 1

Chemistry, 22.06.2019 10:50
An atom of lithium-7 has an equal number of(1) electrons and neutrons(2) electrons and protons(3) positrons and neutrons(4) positrons and protons
Answers: 2
You know the right answer?
Which of the following elements will have its valence electrons represented as in Figure A?
I. Oxyg...
Questions




English, 25.11.2021 06:20




Health, 25.11.2021 06:20

Mathematics, 25.11.2021 06:20


Biology, 25.11.2021 06:20


English, 25.11.2021 06:20

English, 25.11.2021 06:20





Social Studies, 25.11.2021 06:20



