Chemistry, 31.03.2021 07:20 allisonmareesanders2
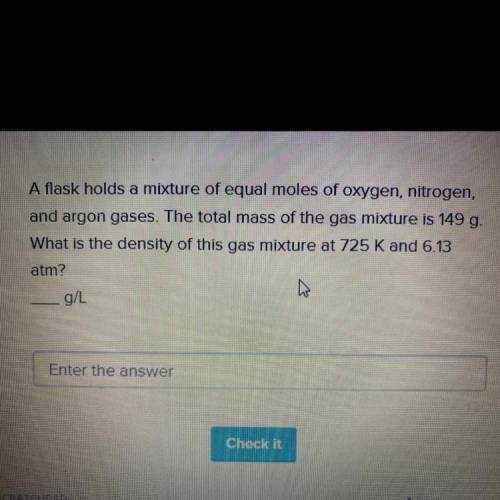
Urgent!! Please help! A flask holds a mixture of equal moles of oxygen, nitrogen, and argon gases. The total mass of the gas mixture is 149 g. What is the density of this gas mixture at 725 K and 6.13 atm?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3

Chemistry, 23.06.2019 07:20
Which statement explains which component is likely to be more powerful in explaining a scientific phenomenon? a) component c, because a theory is often passed on possibility and not certainty b) component d, because a hypothesis is often based on possibility not certainty c) component c, because the ability to explain several occurrences in the natural world is a characteristic of a hypothesis d) component d, because the ability to explain several occurrences in the natural world is a characteristic of a theory
Answers: 3

Chemistry, 23.06.2019 15:40
Glucose (c6h12o6) is the simple sugar that plants make. what is the total number of atoms in glucose? 1 3 24 144
Answers: 1
You know the right answer?
Urgent!! Please help!
A flask holds a mixture of equal moles of oxygen, nitrogen, and argon gases....
Questions

Mathematics, 08.07.2019 17:30


Computers and Technology, 08.07.2019 17:30


Biology, 08.07.2019 17:30

Social Studies, 08.07.2019 17:30


Mathematics, 08.07.2019 17:30

Mathematics, 08.07.2019 17:30

Mathematics, 08.07.2019 17:30


History, 08.07.2019 17:30


English, 08.07.2019 17:30


Mathematics, 08.07.2019 17:30


Mathematics, 08.07.2019 17:30

Mathematics, 08.07.2019 17:30

Mathematics, 08.07.2019 17:30