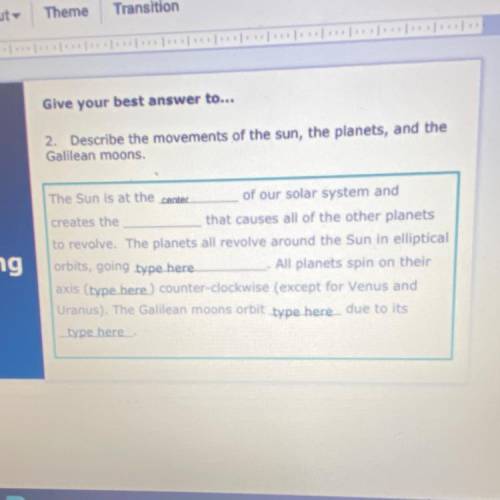
Please help it’s due at 11:59 please
...

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Reaction rate depends on how many molecules are coming into contact with each other with enough energy to react. increasing the temperature of the reactants will increase -
Answers: 3

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2


Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1
You know the right answer?
Questions

Mathematics, 22.05.2021 04:10


Mathematics, 22.05.2021 04:10


History, 22.05.2021 04:10

Mathematics, 22.05.2021 04:10

Mathematics, 22.05.2021 04:10


Mathematics, 22.05.2021 04:10

English, 22.05.2021 04:10




Mathematics, 22.05.2021 04:10



Chemistry, 22.05.2021 04:10


Social Studies, 22.05.2021 04:10

Arts, 22.05.2021 04:10