
Chemistry, 30.03.2021 15:50 jfitness11
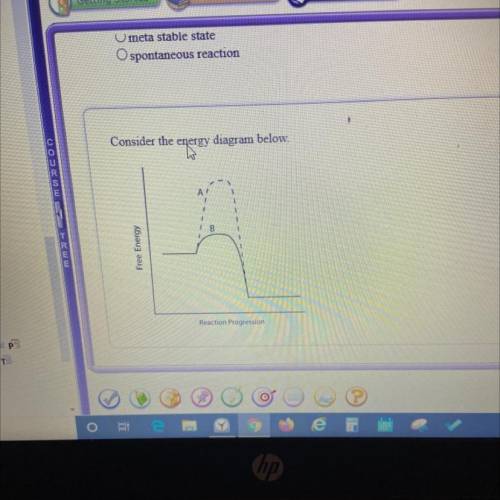
Which line indicates a higher reaction rate?
A. A because it has a lower activation energy.
B. B because it has a lower activation energy.
C. A because its Arxn is much lower.
D. B because its AGеxn is much lower.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 13:30
Table sugar completely dissolved in water is an example of a?
Answers: 1

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 16:50
Ajet plane is speeding down the runway during takeoff. air resistance is not negligible. identify the forces on the jet.
Answers: 3

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1
You know the right answer?
Which line indicates a higher reaction rate?
A. A because it has a lower activation energy.
Questions


Social Studies, 23.08.2019 15:30

English, 23.08.2019 15:30

Health, 23.08.2019 15:30

Social Studies, 23.08.2019 15:30

Mathematics, 23.08.2019 15:30

Mathematics, 23.08.2019 15:30



History, 23.08.2019 15:30

History, 23.08.2019 15:30


Health, 23.08.2019 15:30

History, 23.08.2019 15:30

History, 23.08.2019 15:30

Mathematics, 23.08.2019 15:30






