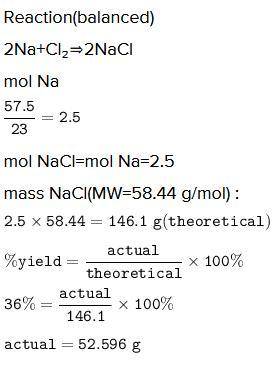Chemistry, 29.03.2021 19:20 joyceslater16
A chemist reacted 57.50 grams of sodium metal with an excess amount of chlorine gas. The chemical reaction that occurred is shown.
Na + Cl2 → NaCl
If the percentage yield of the reaction is 86%, what is the actual yield? Show your work, including the use of stoichiometric calculations and conversion factors.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2

Chemistry, 22.06.2019 13:00
How many moles of sulfur dioxide are produced when 4.38 moles of oxygen completely react with iron (iv) sulfide
Answers: 2

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 22.06.2019 21:30
What is the correct name for the compound cocl3? a) cobalt(i) chloride b) cobalt(i) chlorate c) cobalt(ii) chlorate d) cobalt(iii) chloride
Answers: 1
You know the right answer?
A chemist reacted 57.50 grams of sodium metal with an excess amount of chlorine gas. The chemical re...
Questions


Mathematics, 30.05.2020 19:00


Mathematics, 30.05.2020 19:00

Mathematics, 30.05.2020 19:00





History, 30.05.2020 19:00





Computers and Technology, 30.05.2020 19:00




Mathematics, 30.05.2020 19:01

Mathematics, 30.05.2020 19:01