HELP 10pts!
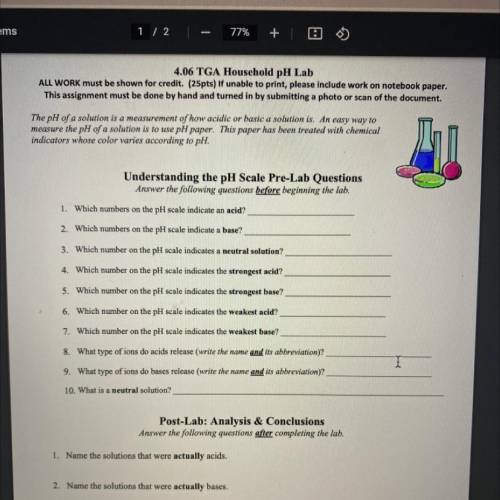
4.06 TGA Household pH Lab
ALL WORK must be shown for credit. (25pts) If unable to...

Chemistry, 25.03.2021 21:20 valoiserika1229
HELP 10pts!
4.06 TGA Household pH Lab
ALL WORK must be shown for credit. (25pts) If unable to print, please include work on notebook paper.
This assignment must be done by hand and turned in by submitting a photo or scan of the document.
The pH of a solution is a measurement of how acidic or basic a solution is. An easy way to
measure the pH of a solution is to use pH paper. This paper has been treated with chemical
indicators whose color varies according to pH.
Understanding the pH Scale Pre-Lab Questions
Answer the following questions before beginning the lab.
1. Which numbers on the pH scale indicate an acid?
2. Which numbers on the pH scale indicate a base?
3. Which number on the pH scale indicates a neutral solution?
4. Which number on the pH scale indicates the strongest acid?
5. Which number on the pH scale indicates the strongest base?
1
6. Which number on the pH scale indicates the weakest acid?
7. Which number on the pH scale indicates the weakest base?
8. What type of ions do acids release (write the name and its abbreviation)?
9. What type of ions do bases release (write the name and its abbreviation)?
10. What is a neutral solution?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 09:40
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1

Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1

Chemistry, 22.06.2019 22:00
Choose all the answers that apply. fluorine (f) has an atomic number of 9 and an atomic weight of 18.99. fluorine has a. 9 protons b. 10 neutrons c. 18 electrons d. an atomic mass of 19 e. at least one isotope
Answers: 1
You know the right answer?
Questions


Social Studies, 23.07.2019 14:00

Biology, 23.07.2019 14:00

Computers and Technology, 23.07.2019 14:00


Social Studies, 23.07.2019 14:00

History, 23.07.2019 14:00

History, 23.07.2019 14:00


Biology, 23.07.2019 14:00

Chemistry, 23.07.2019 14:00






Computers and Technology, 23.07.2019 14:00

Advanced Placement (AP), 23.07.2019 14:00

Social Studies, 23.07.2019 14:00



