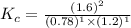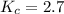Hydrogen and chlorine react to form hydrogen chloride, like this:
H2(g) + Cl,(g) → 2 HCl(g)
...

Chemistry, 24.03.2021 22:30 Kelseygrace8372
Hydrogen and chlorine react to form hydrogen chloride, like this:
H2(g) + Cl,(g) → 2 HCl(g)
Also, a chemist finds that at a certain temperature the equilibrium mixture of hydrogen, chlorine, and hydrogen chloride has the following composition:
compound pressure at equilibrium
H2 0.78
Cl2 1.2M
HCl 1.6M
Calculate the value of the equilibrium constant for this reaction. Round your answer to significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:10
Answer from each drop-down menu. e characteristics of a borane molecule (bh). the lewis structure and table of electronegativities are given olecular shape is and the molecule is reset next erved. search e a
Answers: 2

Chemistry, 22.06.2019 07:40
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na,so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical systems? a) water dissolves nonpolar ionic compounds. b) water dissociates ionic compounds. c) water dissociates covalent molecules. d) water dissolves nonpolar covalent substances.
Answers: 1

Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2
You know the right answer?
Questions

History, 25.11.2021 14:00

History, 25.11.2021 14:00


Mathematics, 25.11.2021 14:00










Spanish, 25.11.2021 14:00

Chemistry, 25.11.2021 14:00





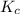
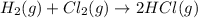
![K_c=\frac{[HCl]^2}{[H_2]^1[I_2]^1}](/tpl/images/1218/2768/5d189.png)