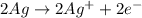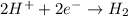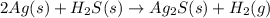2Ag (s) + H2S(s) → Ag2S(s) + H2(g)
Oxidation Half-Reaction:
Reduction Half-Reaction:...

Chemistry, 23.03.2021 06:00 25yurasekth33
2Ag (s) + H2S(s) → Ag2S(s) + H2(g)
Oxidation Half-Reaction:
Reduction Half-Reaction:

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3

Chemistry, 22.06.2019 20:00
Listenbase your answer to the question on the information below.nuclear radiation is harmful to living cells, particularly to fast-growing cells, such as cancer cells and blood cells. an external beam of the radiation emitted from a radioisotope can be directed on a small area of a person to destroy cancer cells within the body.cobalt-60 is an artificially produced radioisotope that emits gamma rays and beta particles. one hospital keeps a 100.0-gram sample of cobalt-60 in an appropriate, secure storage container for future cancer treatment.which choice represents the correct product for the beta decay of the co-60? fe-60ni-60fe-61ni-61
Answers: 2

Chemistry, 23.06.2019 04:40
Temperature is defined as a. the equivalent of heat. b. a measure of the average kinetic energy of the individual atoms or molecules composing a substance. c. how hot or cold it is. d. the total kinetic energy of the atoms or molecules composing a substance. e. none of the above is correct.
Answers: 1

Chemistry, 23.06.2019 05:50
What are the coefficients to balance the following equation? ba+br=babr2
Answers: 1
You know the right answer?
Questions

English, 29.09.2019 06:00


Mathematics, 29.09.2019 06:00


Mathematics, 29.09.2019 06:00


History, 29.09.2019 06:00


Health, 29.09.2019 06:00

Chemistry, 29.09.2019 06:00



Chemistry, 29.09.2019 06:00


Mathematics, 29.09.2019 06:10

Social Studies, 29.09.2019 06:10

Mathematics, 29.09.2019 06:10

English, 29.09.2019 06:10

Social Studies, 29.09.2019 06:10

Mathematics, 29.09.2019 06:10