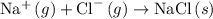Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 13:20
Determine which intermolecular forces are the dominant (strongest) forces for a pure sample of each of the following molecules by placing the molecules into the correct bins. drag the appropriate molecular formula to their respective bins.
Answers: 3

Chemistry, 22.06.2019 09:30
Right anwser gets marked brainliest newton's discovery concerning how fast an object will change speed is the: 1st law 2nd law 3rd law universal gravitation
Answers: 1

Chemistry, 22.06.2019 10:10
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3
You know the right answer?
Write the chemical equation that represents the process of lattice energy for the case of nacl....
Questions

History, 18.03.2020 04:14

Mathematics, 18.03.2020 04:14


Mathematics, 18.03.2020 04:14

Biology, 18.03.2020 04:14

Mathematics, 18.03.2020 04:14



English, 18.03.2020 04:14


History, 18.03.2020 04:15



Mathematics, 18.03.2020 04:15





Mathematics, 18.03.2020 04:16

History, 18.03.2020 04:16

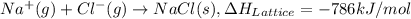
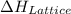 .
.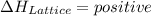 , energy is absorbed while forming of the lattice
, energy is absorbed while forming of the lattice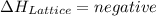 , energy is release while forming of the lattice
, energy is release while forming of the lattice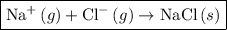
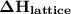 . It can have positive as well as negative values.
. It can have positive as well as negative values.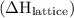 comes out to be positive if the energy supplied to the system is more than that released during the reaction. In other words,
comes out to be positive if the energy supplied to the system is more than that released during the reaction. In other words, 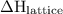 is positive in case of endothermic reactions.
is positive in case of endothermic reactions.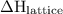 is negative in case of exothermic reactions.
is negative in case of exothermic reactions.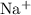 ion and one
ion and one 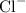 ion. The value of its lattice energy
ion. The value of its lattice energy 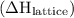 is -787 kJ/mol. A high negative value of lattice energy indicates that the formation of NaCl is highly favored.
is -787 kJ/mol. A high negative value of lattice energy indicates that the formation of NaCl is highly favored.