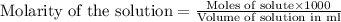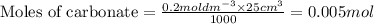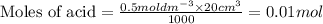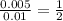A volume of 25cm3 of a carbonate solution of concentration 0.2mol dm-3 was neutralized by 20 cm3 of acid of concentration 0.5 mol dm-3. The mole ratio in which the reactants combine is?
1 mol of carbonate to 1 mol of acid
1 mol of carbonate to 2 mol of acid
2 mol carbonate to 1 mol of acid
3 mol of carbonate to 1 mol of acid

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:10
When will le chatelier's principle come into effect? at the beginning of a reaction, when there are only reactants when a reaction has reached chemical equilibrium when a catalyst is added to a reaction mixture when a reaction is occurring but not yet at equilibrium
Answers: 3

Chemistry, 22.06.2019 03:10
The covalent compound acetylene, which is the fuel of the oxyacetylene torch used by welders, has the molecular formula c2h2. the covalent compound benzene, a commercial solvent, has the molecular formula c6h6 each of these covalent compounds contains carbon and hydrogen atoms in a one-to-one ratio. would it be correct to write the chemical formulas of each as ch? explain.
Answers: 1

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 11:00
What is the temperature of 0.750 mol of a gas stored in a 6,850 ml cylinder at 2.21 atm? . 2.95 k 5.24 k 138 k 246 k
Answers: 3
You know the right answer?
A volume of 25cm3 of a carbonate solution of concentration 0.2mol dm-3 was neutralized by 20 cm3 of...
Questions

Social Studies, 18.01.2022 03:10

English, 18.01.2022 03:10



Mathematics, 18.01.2022 03:10

Mathematics, 18.01.2022 03:10

Social Studies, 18.01.2022 03:10

Mathematics, 18.01.2022 03:10

History, 18.01.2022 03:10

Mathematics, 18.01.2022 03:10

Mathematics, 18.01.2022 03:10


English, 18.01.2022 03:10



Mathematics, 18.01.2022 03:10



Mathematics, 18.01.2022 03:10

Social Studies, 18.01.2022 03:10