
Potassium chlorate, a common oxidizing agent in fireworks and matchheads, undergoes a solid-state disproportionation reaction when heated:
4KClO3 (s) ⟶ Δ3KClO4 (s) + KCl (s).
Use ΔHf ° and S° values to calculate ΔG_sys ° (which is ΔGrxn °) in kJ at 25°C for this reaction.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
One does not belong why? ice, gold ,wood ,diamond and table salt
Answers: 1


Chemistry, 22.06.2019 14:00
Ascientist measures the speed of sound in a monatomic gas to be 449 m/s at 20∘c. what is the molar mass of this gas?
Answers: 2

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3
You know the right answer?
Potassium chlorate, a common oxidizing agent in fireworks and matchheads, undergoes a solid-state di...
Questions


Physics, 03.02.2020 15:52

Mathematics, 03.02.2020 15:52



Mathematics, 03.02.2020 15:52






History, 03.02.2020 15:52

History, 03.02.2020 15:52

Mathematics, 03.02.2020 15:52

Mathematics, 03.02.2020 15:52

Health, 03.02.2020 15:52



Physics, 03.02.2020 15:52

Mathematics, 03.02.2020 15:52

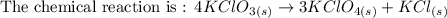

![\implies G^0_{rxn} = 3 \times \Delta _fG^0 [KClO_4{(s)}] + \Delta_fG^0[KCl_{(s)}] - 4 \times \Delta _f G^0 [ KClO_3 (s) ]](/tpl/images/1206/8925/2b60b.png)
![\Delta _fG^0 \ values \ at \ 25^0 \ C (298 \ K) are\ given \ as:\\\\ \Delta _fG^0 [KClO_4(s)] = -303.09 \ kJ \\ \\ \Delta _fG^0 [KCl(s) ] = - 409.14 \ kJ \\ \\ \Delta_f G^0 [KClO_3_{(s)}] = -296.25 \ kJ \\ \\ replacing \ the \ above \ values \ into \ equation (1) ; then:\\ \\ \\\Delta G^0_{rxn} = 3 *(-303.09) + (-409.14) - 4*(-296.25) \ kJ \\ \\ = (-909.27 - 409.14 + 1185) \ kJ \\ \\ = -133.41 \ kJ \\ \\ \mathbf{\Delta G^0_{rxn} = -133.4 \ kJ }](/tpl/images/1206/8925/f4981.png)


