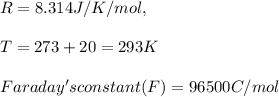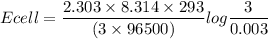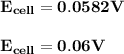A certain metal M forms a soluble nitrate salt M NO Suppose the left half cell of a galvanic cell apparatus is filled with a 3.00 mM solution of M(NO) and the right half cell with a 3.00 M solution of the same substance. Electrodes made of M are dipped into both solutions and a voltmeter is connected between them. The temperature of the apparatus is held constant at 20.0 °C.
1. Which electrode will be positive? What voltage will the voltmeter show?
a. left
b. right
2. Assume its positive lead is connected to the positive electrode. Be sure your answer has a unit symbol, if necessary, and round it to 2 significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:00
What mass of carbon dioxide is produced from the complete combustion of 4.50×10−3 g of methane?
Answers: 2

Chemistry, 22.06.2019 13:30
What does the xylem do? stores the glucose captures the sunlight absorbs oxygen into the leaf carries water from the roots to the leaves
Answers: 1

Chemistry, 22.06.2019 16:00
No copying 15 pts how does a free-body diagram tell you about the net force on an object?
Answers: 2

Chemistry, 22.06.2019 20:00
How are the terms group and period used on the periodic table
Answers: 1
You know the right answer?
A certain metal M forms a soluble nitrate salt M NO Suppose the left half cell of a galvanic cell ap...
Questions

Mathematics, 23.10.2020 05:01

Biology, 23.10.2020 05:01





Mathematics, 23.10.2020 05:01



Health, 23.10.2020 05:01

Computers and Technology, 23.10.2020 05:01

Mathematics, 23.10.2020 05:01

Mathematics, 23.10.2020 05:01



Mathematics, 23.10.2020 05:01


Mathematics, 23.10.2020 05:01


Mathematics, 23.10.2020 05:01

![Ecell =\Big( \dfrac{2.303\times R\times T}{nF} \Big)log\Big(\dfrac{[cathode]}{[anode]}\Big)](/tpl/images/1206/7747/f3d33.png)