
Which is the Net Ionic Equation for:
(remember, the Net Ionic Eq. shows ALL of the PARTICIPANTS)
2FeCl3(aq) + 3Mg(s) ® 3MgCl2(aq) + 2Fe(s)
2Fe3+(aq) + 3Mg(s) ® 3Mg2+(s) + 2Fe(aq)
2Fe3+ (aq) + Cl3- (aq) + 3Mg(s) ® 3Mg3+ (aq) + Cl2- (aq) + 2Fe(s)
2Fe3+ (aq) + Cl3- (aq) + 3Mg(s) ® 3Mg3+ (aq) + Cl2- (aq) + 2Fe(s)
2Fe3+ (aq) + 6Cl- (aq) + 3Mg(s) ® 3Mg2+ (aq) + 6Cl- (aq) + 2Fe(s)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1



Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2
You know the right answer?
Which is the Net Ionic Equation for:
(remember, the Net Ionic Eq. shows ALL of the PARTICIPANTS)
Questions

English, 05.10.2019 03:30

Mathematics, 05.10.2019 03:30

Physics, 05.10.2019 03:30


Mathematics, 05.10.2019 03:30

Spanish, 05.10.2019 03:30

Mathematics, 05.10.2019 03:30


Arts, 05.10.2019 03:30



Biology, 05.10.2019 03:30


Mathematics, 05.10.2019 03:30

Biology, 05.10.2019 03:30

Mathematics, 05.10.2019 03:30

History, 05.10.2019 03:30



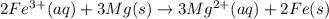
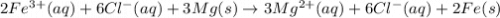
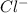 are the spectator ions.
are the spectator ions.


