

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 21.06.2019 18:00
What is the theoretical yield of carbon dioxide? a)0.993 gb)2.98 gc)3.65 gd)8.93 g
Answers: 1

Chemistry, 21.06.2019 22:30
Joseph has hypothesized that sound travels in waves. if he were following the scientific method, what should he do next? a. ask a question. b. test the hypothesis. c. study the results. d. tell other scientists about his hypothesis.
Answers: 1

Chemistry, 22.06.2019 03:00
Which of the dna typing techniques do you think you would choose if you had to analyze a dna sample? why?
Answers: 1
You know the right answer?
The concentration of Mg2+ in seawater is 0.052 M. At what pH will 76% of the Mg2+ be precipitated as...
Questions


History, 03.03.2021 23:00

History, 03.03.2021 23:00

History, 03.03.2021 23:00




Mathematics, 03.03.2021 23:00

Spanish, 03.03.2021 23:00

Computers and Technology, 03.03.2021 23:00


Social Studies, 03.03.2021 23:00

Mathematics, 03.03.2021 23:00







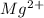 will be precipitated as the hydroxide salt.
will be precipitated as the hydroxide salt.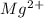 has precipitated out , 24% remains out.
has precipitated out , 24% remains out.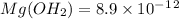
![[Mg^{2}^{+}] =\frac{24}{100}\times 0.052](/tpl/images/1200/9004/3cfbf.png)

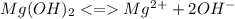
![Ksp = [Mg^2^+][OH^-]](/tpl/images/1200/9004/4a0be.png)
![8.9\times 10^-^1^2= 1.248\times10^-^2\times[OH]^-](/tpl/images/1200/9004/8a6a7.png)
![[OH]^- = 7.131\times 10^-^1^0](/tpl/images/1200/9004/e943a.png)
![[H^+]=\frac{Kw}{[OH]^-}](/tpl/images/1200/9004/234a2.png) ( where Kw is the ionic product of the water)
( where Kw is the ionic product of the water)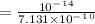

![pH=-Log[H^+]](/tpl/images/1200/9004/3ca39.png)
![-Log[1.402\times10^-^5]](/tpl/images/1200/9004/c76b6.png)


