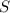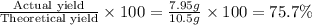2). H2S and SO2 as follows,(8pt)
H2S + SO2 >S + H2O
In a particular experiment, 7.9...

Chemistry, 18.03.2021 01:50 reginaldboyd28
2). H2S and SO2 as follows,(8pt)
H2S + SO2 >S + H2O
In a particular experiment, 7.95 g of sulfur is produced by the reaction of 7.5 g of H2S with 12.75 g of SO2.
What is the % yield of sulfur?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1


Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 3
You know the right answer?
Questions




English, 22.10.2020 17:01

Mathematics, 22.10.2020 17:01



History, 22.10.2020 17:01

Mathematics, 22.10.2020 17:01

English, 22.10.2020 17:01


Mathematics, 22.10.2020 17:01

Mathematics, 22.10.2020 17:01

Mathematics, 22.10.2020 17:01

Mathematics, 22.10.2020 17:01






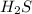 will require = 64 g of
will require = 64 g of 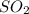
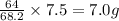 of
of 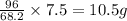 of
of