
Chemistry, 17.03.2021 23:50 JamesLachoneus
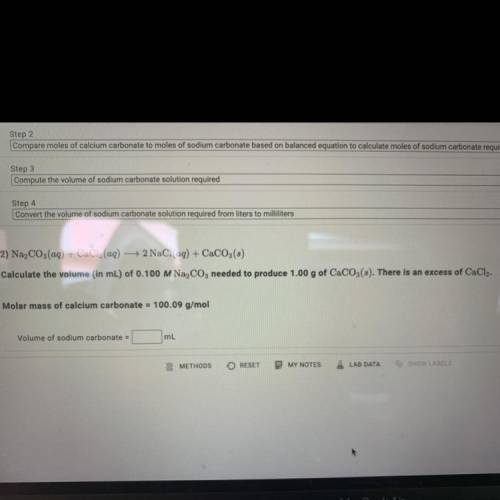
Calculate the volume (in mL) of 0.100 M Na, C03 needed to produce 1.00 g of CaCO3(s).
There is an excess of CaCl2.
Molar mass of calcium carbonate = 100.09 g/mol
Volume of sodium carbonate = ?mL

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
The compound methyl butanoate smells like apples. its percent composition is 58.8% c, 9.9% h, and 31.4% o. what’s the empirical formula ?
Answers: 1

Chemistry, 21.06.2019 22:30
Hot air balloons float in the air because of the difference in density between cold and hot air. in this problem, you will estimate the minimum temperature the gas inside the balloon needs to be, for it to take off. to do this, use the following variables and make these assumptions: the combined weight of the pilot basket together with that of the balloon fabric and other equipment is w. the volume of the hot air inside the balloon when it is inflated is v. the absolute temperature of the hot air at the bottom of the balloon is th (where th> tc). the absolute temperature of the cold air outside the balloon is tc and its density is ďc. the balloon is open at the bottom, so that the pressure inside and outside the balloon is the same. as always, treat air as an ideal gas. use g for the magnitude of the acceleration due to gravity.
Answers: 1

Chemistry, 22.06.2019 12:00
Which of the following units is not an official si unit? mole liter kilogram ampere
Answers: 1

Chemistry, 23.06.2019 01:00
How does carbon monoxide pose the greatest threat to humans? a. it can be produced by wood fires. b. it can be produced by home furnaces. c. it is produced by acid rain. d. it is produced by modern automobiles.
Answers: 2
You know the right answer?
Calculate the volume (in mL) of 0.100 M Na, C03 needed to produce 1.00 g of CaCO3(s).
There is an e...
Questions

English, 01.08.2019 12:10

Mathematics, 01.08.2019 12:10








Mathematics, 01.08.2019 12:10



Chemistry, 01.08.2019 12:20




Advanced Placement (AP), 01.08.2019 12:20

Mathematics, 01.08.2019 12:20




