
Chemistry, 17.03.2021 23:50 bella122805
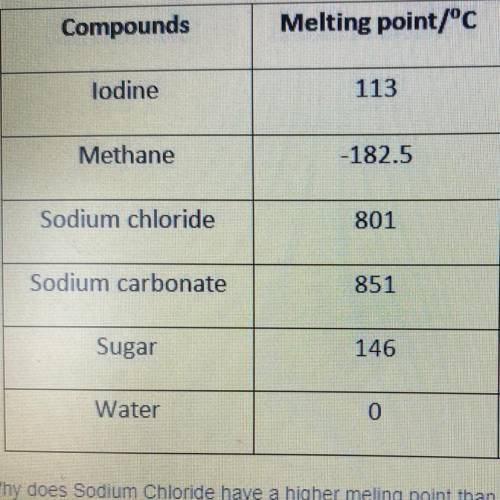
Why does Sodium Chloride have a higher melting point than Sugar?
a. intermolecular forces are weaker
b. intermolecular forces are slightly stronger
c. intermolecular forces are very strong
d. melting point is based on composition and not bonding

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 16:20
Consider the two electron arrangements for neutral atoms a and b. are atoms a and b the same element? a - 1s2, 2s2, 2p6, 3s1 b - 1s2, 2s2, 2p6, 5s1
Answers: 3

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 17:30
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1

Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2
You know the right answer?
Why does Sodium Chloride have a higher melting point than Sugar?
a. intermolecular forces are weake...
Questions




Mathematics, 20.11.2020 14:00





Social Studies, 20.11.2020 14:00

Computers and Technology, 20.11.2020 14:00

Mathematics, 20.11.2020 14:00







Mathematics, 20.11.2020 14:00

Computers and Technology, 20.11.2020 14:00



